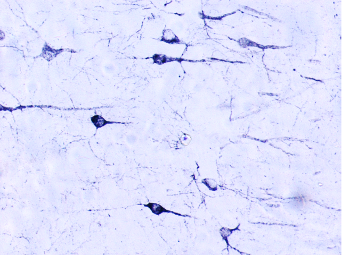
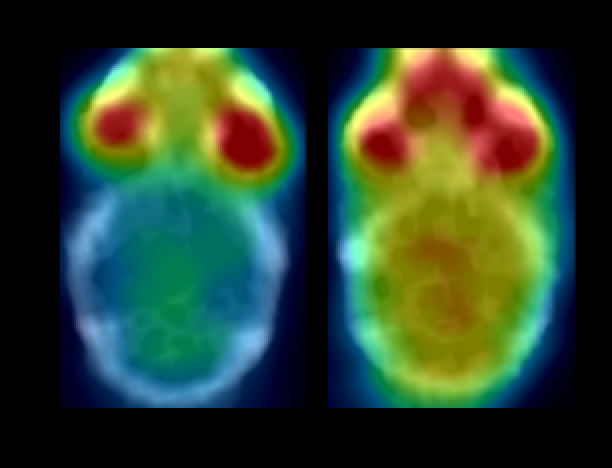
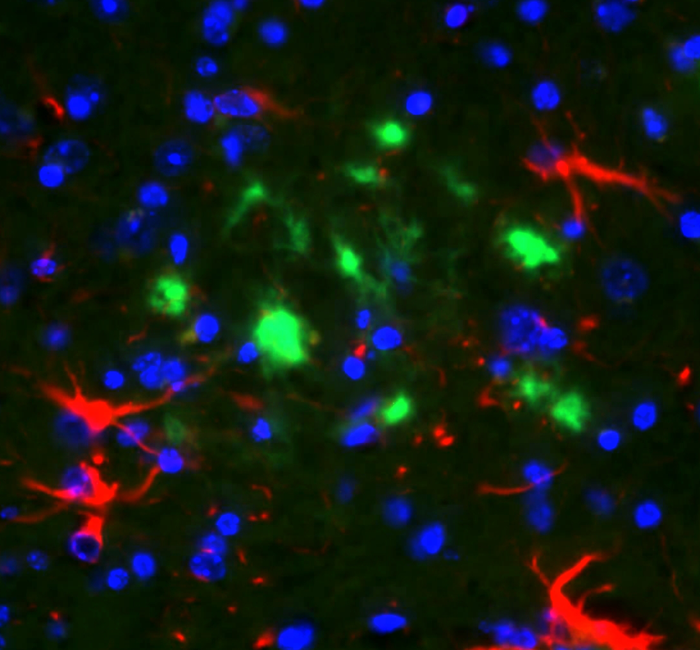
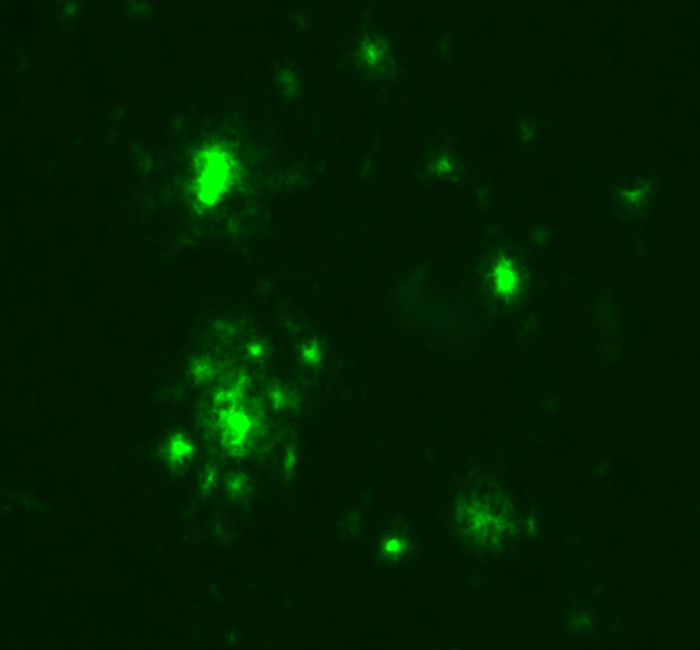
The progression of tau pathology in the brain of Alzheimer’s patients correlates with the neurodegenerative process and cognitive decline. Under experimental conditions, tau aggregates can spread and accelerate the progression of tauopathies. Moreover, it is possible to modify the levels of beta-amyloid in the brain by changing its balance in the periphery. Our aim is to explore the role of vascular tau aggregates in the development or treatment of the disease.
Funded by Ministerio de Ciencia e Innovación